Tecfidera Prescribing Information
Package insert / product label
Generic name: dimethyl fumarate
Dosage form: capsules
Drug class: Selective immunosuppressants
Medically reviewed by Drugs.com. Last updated on Mar 25, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
TECFIDERA® (dimethyl fumarate) delayed-release capsules, for oral use
Initial U.S. Approval: 2013
Recent Major Changes
| Warnings and Precautions, Serious Gastrointestinal Reactions (5.7) | 12/2023 |
Indications and Usage for Tecfidera
TECFIDERA is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults. (1)
Tecfidera Dosage and Administration
Dosage Forms and Strengths
Delayed-release capsules: 120 mg and 240 mg (3)
Contraindications
Known hypersensitivity to dimethyl fumarate or any of the excipients of TECFIDERA. (4)
Warnings and Precautions
- Anaphylaxis and Angioedema: Discontinue and do not restart TECFIDERA if these occur. (5.1)
- Progressive Multifocal Leukoencephalopathy (PML): Withhold TECFIDERA at the first sign or symptom suggestive of PML. (5.2)
- Herpes Zoster and Other Serious Opportunistic Infections: Consider withholding TECFIDERA in cases of serious infection until the infection has resolved. (5.3)
- Lymphopenia: Obtain a CBC including lymphocyte count before initiating TECFIDERA, after 6 months, and every 6 to 12 months thereafter. Consider interruption of TECFIDERA if lymphocyte counts <0.5 x 109/L persist for more than six months. (5.4)
- Liver Injury: Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels before initiating TECFIDERA and during treatment, as clinically indicated. Discontinue TECFIDERA if clinically significant liver injury induced by TECFIDERA is suspected. (5.5)
Adverse Reactions/Side Effects
Most common adverse reactions (incidence ≥10% and ≥2% placebo) were flushing, abdominal pain, diarrhea, and nausea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Biogen at 1-800-456-2255 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2024
Full Prescribing Information
1. Indications and Usage for Tecfidera
TECFIDERA is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
2. Tecfidera Dosage and Administration
2.1 Dosing Information
The starting dose for TECFIDERA is 120 mg twice a day orally. After 7 days, the dose should be increased to the maintenance dose of 240 mg twice a day orally. Temporary dose reductions to 120 mg twice a day may be considered for individuals who do not tolerate the maintenance dose. Within 4 weeks, the recommended dose of 240 mg twice a day should be resumed. Discontinuation of TECFIDERA should be considered for patients unable to tolerate return to the maintenance dose. The incidence of flushing may be reduced by administration of TECFIDERA with food. Alternatively, administration of non-enteric coated aspirin (up to a dose of 325 mg) 30 minutes prior to TECFIDERA dosing may reduce the incidence or severity of flushing [see Clinical Pharmacology (12.3)].
TECFIDERA should be swallowed whole and intact. TECFIDERA should not be crushed or chewed, and the capsule contents should not be sprinkled on food. TECFIDERA can be taken with or without food.
2.2 Blood Tests Prior to Initiation of Therapy
Obtain a complete blood cell count (CBC) including lymphocyte count before initiation of therapy [see Warnings and Precautions (5.4)].
Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels prior to treatment with TECFIDERA [see Warnings and Precautions (5.5)].
3. Dosage Forms and Strengths
TECFIDERA is available as hard gelatin delayed-release capsules containing 120 mg or 240 mg of dimethyl fumarate. The 120 mg capsules have a green cap and white body, printed with “BG-12 120 mg” in black ink on the body. The 240 mg capsules have a green cap and a green body, printed with “BG-12 240 mg” in black ink on the body.
4. Contraindications
TECFIDERA is contraindicated in patients with known hypersensitivity to dimethyl fumarate or to any of the excipients of TECFIDERA. Reactions have included anaphylaxis and angioedema [see Warnings and Precautions (5.1)].
5. Warnings and Precautions
5.1 Anaphylaxis and Angioedema
TECFIDERA can cause anaphylaxis and angioedema after the first dose or at any time during treatment. Signs and symptoms have included difficulty breathing, urticaria, and swelling of the throat and tongue. Patients should be instructed to discontinue TECFIDERA and seek immediate medical care should they experience signs and symptoms of anaphylaxis or angioedema.
5.2 Progressive Multifocal Leukoencephalopathy
Progressive multifocal leukoencephalopathy (PML) has occurred in patients with MS treated with TECFIDERA. PML is an opportunistic viral infection of the brain caused by the JC virus (JCV) that typically only occurs in patients who are immunocompromised, and that usually leads to death or severe disability. A fatal case of PML occurred in a patient who received TECFIDERA for 4 years while enrolled in a clinical trial. During the clinical trial, the patient experienced prolonged lymphopenia (lymphocyte counts predominantly <0.5x109/L for 3.5 years) while taking TECFIDERA [see Warnings and Precautions (5.4)]. The patient had no other identified systemic medical conditions resulting in compromised immune system function and had not previously been treated with natalizumab, which has a known association with PML. The patient was also not taking any immunosuppressive or immunomodulatory medications concomitantly.
PML has also occurred in the postmarketing setting in the presence of lymphopenia (<0.9x109/L). While the role of lymphopenia in these cases is uncertain, the PML cases have occurred predominantly in patients with lymphocyte counts <0.8x109/L persisting for more than 6 months.
At the first sign or symptom suggestive of PML, withhold TECFIDERA and perform an appropriate diagnostic evaluation. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes.
MRI findings may be apparent before clinical signs or symptoms. Cases of PML, diagnosed based on MRI findings and the detection of JCV DNA in the cerebrospinal fluid in the absence of clinical signs or symptoms specific to PML, have been reported in patients treated with other MS medications associated with PML. Many of these patients subsequently became symptomatic with PML. Therefore, monitoring with MRI for signs that may be consistent with PML may be useful, and any suspicious findings should lead to further investigation to allow for an early diagnosis of PML, if present. Lower PML-related mortality and morbidity have been reported following discontinuation of another MS medication associated with PML in patients with PML who were initially asymptomatic compared to patients with PML who had characteristic clinical signs and symptoms at diagnosis. It is not known whether these differences are due to early detection and discontinuation of MS treatment or due to differences in disease in these patients.
5.3 Herpes Zoster and Other Serious Opportunistic Infections
Serious cases of herpes zoster have occurred with TECFIDERA, including disseminated herpes zoster, herpes zoster ophthalmicus, herpes zoster meningoencephalitis, and herpes zoster meningomyelitis. These events may occur at any time during treatment. Monitor patients on TECFIDERA for signs and symptoms of herpes zoster. If herpes zoster occurs, appropriate treatment for herpes zoster should be administered.
Other serious opportunistic infections have occurred with TECFIDERA, including cases of serious viral (herpes simplex virus, West Nile virus, cytomegalovirus), fungal (Candida and Aspergillus), and bacterial (Nocardia, Listeria monocytogenes, Mycobacterium tuberculosis) infections. These infections have been reported in patients with reduced absolute lymphocyte counts (ALC) as well as in patients with normal ALC. These infections have affected the brain, meninges, spinal cord, gastrointestinal tract, lungs, skin, eye, and ear. Patients with symptoms and signs consistent with any of these infections should undergo prompt diagnostic evaluation and receive appropriate treatment.
Consider withholding TECFIDERA treatment in patients with herpes zoster or other serious infections until the infection has resolved [see Adverse Reactions (6.2)].
5.4 Lymphopenia
TECFIDERA may decrease lymphocyte counts. In the MS placebo controlled trials, mean lymphocyte counts decreased by approximately 30% during the first year of treatment with TECFIDERA and then remained stable. Four weeks after stopping TECFIDERA, mean lymphocyte counts increased but did not return to baseline. Six percent (6%) of TECFIDERA patients and <1% of placebo patients experienced lymphocyte counts <0.5x109/L (lower limit of normal 0.91x109/L). The incidence of infections (60% vs 58%) and serious infections (2% vs 2%) was similar in patients treated with TECFIDERA or placebo, respectively. There was no increased incidence of serious infections observed in patients with lymphocyte counts <0.8x109/L or ≤0.5x109/L in controlled trials, although one patient in an extension study developed PML in the setting of prolonged lymphopenia (lymphocyte counts predominantly <0.5x109/L for 3.5 years) [see Warnings and Precautions (5.2)].
In controlled and uncontrolled clinical trials, 2% of patients experienced prolonged, severe lymphopenia, (defined as lymphocyte counts <0.5 x 109/L for at least six months); in this group of patients, the majority of lymphocyte counts remained <0.5x109/L with continued therapy. In these patients with prolonged, severe lymphopenia, the median time for lymphocyte counts to return to normal after discontinuing TECFIDERA was 96.0 weeks.
In these controlled and uncontrolled clinical studies, among patients who did not experience prolonged, severe lymphopenia during treatment, the median times for lymphocyte counts to return to normal after discontinuing TECFIDERA were as follows:
- 4.3 weeks in patients with mild lymphopenia (lymphocyte count ≥0.8x109/L) at discontinuation,
- 10.0 weeks in patients with moderate lymphopenia (lymphocyte count 0.5 to <0.8x109/L) at discontinuation, and
- 16.7 weeks in patients with severe lymphopenia (lymphocyte count <0.5x109/L) at discontinuation.
TECFIDERA has not been studied in patients with pre-existing low lymphocyte counts.
Obtain a CBC, including lymphocyte count, before initiating treatment with TECFIDERA, 6 months after starting treatment, and then every 6 to 12 months thereafter, and as clinically indicated. Consider interruption of TECFIDERA in patients with lymphocyte counts less than 0.5 x 109/L persisting for more than six months. Given the potential for delayed recovery of lymphocyte counts, continue to obtain lymphocyte counts until their recovery if TECFIDERA is discontinued or interrupted due to lymphopenia. Consider withholding treatment from patients with serious infections until resolution. Decisions about whether or not to restart TECFIDERA should be individualized based on clinical circumstances.
5.5 Liver Injury
Clinically significant cases of liver injury have been reported in patients treated with TECFIDERA in the postmarketing setting. The onset has ranged from a few days to several months after initiation of treatment with TECFIDERA. Signs and symptoms of liver injury, including elevation of serum aminotransferases to greater than 5-fold the upper limit of normal and elevation of total bilirubin to greater than 2-fold the upper limit of normal have been observed. These abnormalities resolved upon treatment discontinuation. Some cases required hospitalization. None of the reported cases resulted in liver failure, liver transplant, or death. However, the combination of new serum aminotransferase elevations with increased levels of bilirubin caused by drug-induced hepatocellular injury is an important predictor of serious liver injury that may lead to acute liver failure, liver transplant, or death in some patients.
Elevations of hepatic transaminases (most no greater than 3 times the upper limit of normal) were observed during controlled trials [see Adverse Reactions (6.1)].
Obtain serum aminotransferase, alkaline phosphatase (ALP), and total bilirubin levels prior to treatment with TECFIDERA and during treatment, as clinically indicated. Discontinue TECFIDERA if clinically significant liver injury induced by TECFIDERA is suspected.
5.6 Flushing
TECFIDERA may cause flushing (e.g., warmth, redness, itching, and/or burning sensation). In clinical trials, 40% of TECFIDERA treated patients experienced flushing. Flushing symptoms generally began soon after initiating TECFIDERA and usually improved or resolved over time. In the majority of patients who experienced flushing, it was mild or moderate in severity. Three percent (3%) of patients discontinued TECFIDERA for flushing and <1% had serious flushing symptoms that were not life-threatening but led to hospitalization. Administration of TECFIDERA with food may reduce the incidence of flushing. Alternatively, administration of non-enteric coated aspirin (up to a dose of 325 mg) 30 minutes prior to TECFIDERA dosing may reduce the incidence or severity of flushing [see Dosing and Administration (2.1) and Clinical Pharmacology (12.3)].
5.7 Serious Gastrointestinal Reactions
Serious gastrointestinal reactions, including perforation, ulceration, hemorrhage, and obstruction, some with fatal outcomes, have been reported in the postmarketing setting with the use of fumaric acid esters, including TECFIDERA, with or without concomitant aspirin use. The majority of these events have occurred within 6 months of fumaric acid ester treatment initiation. In controlled clinical trials, the incidence of serious gastrointestinal adverse events was 1% in patients treated with TECFIDERA; these events, none of which were fatal, included vomiting (0.3%) and abdominal pain (0.3%) [see Adverse Reactions (6.1)].
Monitor patients, promptly evaluate, and discontinue TECFIDERA for new or worsening severe gastrointestinal signs and symptoms.
6. Adverse Reactions/Side Effects
The following important adverse reactions are described elsewhere in labeling:
- Anaphylaxis and Angioedema [see Warnings and Precautions (5.1)].
- Progressive multifocal leukoencephalopathy [see Warnings and Precautions (5.2)].
- Herpes Zoster and Other Serious Opportunistic Infections [see Warnings and Precautions (5.3)].
- Lymphopenia [see Warnings and Precautions (5.4)].
- Liver Injury [see Warnings and Precautions (5.5)].
- Flushing [see Warnings and Precautions (5.6)].
- Serious Gastrointestinal Reactions [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In placebo-controlled and uncontrolled clinical studies, a total of 2513 patients have received TECFIDERA and been followed for periods up to 13 years with an overall exposure of 11,318 person-years. Approximately 1169 patients have received more than 5 years of treatment with TECFIDERA, and 426 patients have received at least 10 years of treatment with TECFIDERA.
Adverse Reactions in Placebo-Controlled Trials
In the two well-controlled studies demonstrating effectiveness, 1529 patients received TECFIDERA with an overall exposure of 2244 person-years [see Clinical Studies (14)].
The adverse reactions presented in the table below are based on safety information from 769 patients treated with TECFIDERA 240 mg twice a day and 771 placebo-treated patients.
The most common adverse reactions (incidence ≥10% and ≥2% more than placebo) for TECFIDERA were flushing, abdominal pain, diarrhea, and nausea.
| TECFIDERA
N=769 % | Placebo
N=771 % |
|
| Flushing | 40 | 6 |
| Abdominal pain | 18 | 10 |
| Diarrhea | 14 | 11 |
| Nausea | 12 | 9 |
| Vomiting | 9 | 5 |
| Pruritus | 8 | 4 |
| Rash | 8 | 3 |
| Albumin urine present | 6 | 4 |
| Erythema | 5 | 1 |
| Dyspepsia | 5 | 3 |
| Aspartate aminotransferase increased | 4 | 2 |
| Lymphopenia | 2 | <1 |
Gastrointestinal
TECFIDERA caused GI events (e.g., nausea, vomiting, diarrhea, abdominal pain, and dyspepsia). The incidence of GI events was higher early in the course of treatment (primarily in month 1) and usually decreased over time in patients treated with TECFIDERA compared with placebo. Four percent (4%) of patients treated with TECFIDERA and less than 1% of placebo patients discontinued due to gastrointestinal events. The incidence of serious GI events was 1% in clinical trial patients treated with TECFIDERA; these events, none of which were fatal, included vomiting (0.3%) and abdominal pain (0.3%).
Hepatic Transaminases
An increased incidence of elevations of hepatic transaminases in patients treated with TECFIDERA was seen primarily during the first six months of treatment, and most patients with elevations had levels < 3 times the upper limit of normal (ULN) during controlled trials. Elevations of alanine aminotransferase and aspartate aminotransferase to ≥ 3 times the ULN occurred in a small number of patients treated with both TECFIDERA and placebo and were balanced between groups. There were no elevations in transaminases ≥ 3 times the ULN with concomitant elevations in total bilirubin > 2 times the ULN. Discontinuations due to elevated hepatic transaminases were < 1% and were similar in patients treated with TECFIDERA or placebo.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of TECFIDERA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Acute Pancreatitis; Gastrointestinal perforation, ulceration, obstruction, and hemorrhage [see Warnings and Precautions (5.7)]
Hepatobiliary Disorders: Liver function abnormalities (elevations in transaminases ≥ 3 times ULN with concomitant elevations in total bilirubin > 2 times ULN) [see Warnings and Precautions (5.5)]
Infections and Infestations: Herpes zoster infection and other serious opportunistic infections [see Warnings and Precautions (5.3)]
Respiratory, Thoracic, and Mediastinal Disorders: Rhinorrhea
Skin and Subcutaneous: Alopecia
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Available data from the TECFIDERA Pregnancy Registry, observational studies, and pharmacovigilance with dimethyl fumarate use in pregnant women have not indicated an increased risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Most of the reported exposures to dimethyl fumarate occurred during the first trimester of pregnancy (see Data). In animals, adverse effects on offspring survival, growth, sexual maturation, and neurobehavioral function were observed when dimethyl fumarate (DMF) was administered during pregnancy and lactation at clinically relevant doses (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Human Data
In a prospective observational TECFIDERA Pregnancy Registry (2013-2022), the rate of major birth defects among 362 live births and stillbirths from women who were exposed to dimethyl fumarate during pregnancy was 3.6% (95% CI: 1.9-6.1). No specific pattern of major birth defects was identified. Important potential study limitations include exposure misclassification, no adjustment for confounders, and lack of an internal comparator cohort.
Animal Data
In rats administered DMF orally (25, 100, 250 mg/kg/day) throughout organogenesis, embryofetal toxicity (reduced fetal body weight and delayed ossification) were observed at the highest dose tested. This dose also produced evidence of maternal toxicity (reduced body weight). Plasma exposure (AUC) for monomethyl fumarate (MMF), the major circulating metabolite, at the no-effect dose is approximately three times that in humans at the recommended human dose (RHD) of 480 mg/day. In rabbits administered DMF orally (25, 75, and 150 mg/kg/day) throughout organogenesis, embryolethality and decreased maternal body weight were observed at the highest dose tested. The plasma AUC for MMF at the no-effect dose is approximately 5 times that in humans at the RHD.
Oral administration of DMF (25, 100, and 250 mg/kg/day) to rats throughout organogenesis and lactation resulted in increased lethality, persistent reductions in body weight, delayed sexual maturation (male and female pups), and reduced testicular weight at the highest dose tested. Neurobehavioral impairment was observed at all doses. A no-effect dose for developmental toxicity was not identified. The lowest dose tested was associated with plasma AUC for MMF lower than that in humans at the RHD.
8.2 Lactation
Risk Summary
There are no data on the presence of DMF or MMF in human milk. The effects on the breastfed infant and on milk production are unknown.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for TECFIDERA and any potential adverse effects on the breastfed infant from the drug or from the underlying maternal condition.
10 OVERDOSE
Cases of overdose with TECFIDERA have been reported. The symptoms described in these cases were consistent with the known adverse event profile of TECFIDERA.
There are no known therapeutic interventions to enhance elimination of TECFIDERA nor is there a known antidote. In the event of overdose, initiate symptomatic supportive treatment as clinically indicated.
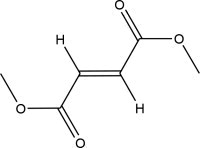
11. Tecfidera Description
TECFIDERA contains dimethyl fumarate which is also known by its chemical name, dimethyl (E) butenedioate, (C6H8O4). It has the following structure:
Dimethyl fumarate is a white to off-white powder that is highly soluble in water with a molecular mass of 144.13.
TECFIDERA is provided as hard gelatin delayed-release capsules for oral administration, containing 120 mg or 240 mg of dimethyl fumarate consisting of the following inactive ingredients: microcrystalline cellulose, silicified microcrystalline cellulose, croscarmellose sodium, talc, silica colloidal silicon dioxide, magnesium stearate, triethyl citrate, methacrylic acid copolymer - Type A, methacrylic acid copolymer dispersion, simethicone (30% emulsion), sodium lauryl sulphate, and polysorbate 80. The capsule shell, printed with black ink, contains the following inactive ingredients: gelatin, titanium dioxide, FD&C blue 1; brilliant blue FCF, yellow iron oxide and black iron oxide.
12. Tecfidera - Clinical Pharmacology
12.1 Mechanism of Action
The mechanism by which dimethyl fumarate (DMF) exerts its therapeutic effect in multiple sclerosis is unknown. DMF and the metabolite, monomethyl fumarate (MMF), have been shown to activate the Nuclear factor (erythroid-derived 2)-like 2 (Nrf2) pathway in vitro and in vivo in animals and humans. The Nrf2 pathway is involved in the cellular response to oxidative stress. MMF has been identified as a nicotinic acid receptor agonist in vitro.
12.2 Pharmacodynamics
Potential to prolong the QT interval
In a placebo controlled thorough QT study performed in healthy subjects, there was no evidence that dimethyl fumarate caused QT interval prolongation of clinical significance (i.e., the upper bound of the 90% confidence interval for the largest placebo-adjusted, baseline-corrected QTc was below 10 ms).
12.3 Pharmacokinetics
After oral administration of TECFIDERA, dimethyl fumarate undergoes rapid presystemic hydrolysis by esterases and is converted to its active metabolite, monomethyl fumarate (MMF). Dimethyl fumarate is not quantifiable in plasma following oral administration of TECFIDERA. Therefore all pharmacokinetic analyses related to TECFIDERA were performed with plasma MMF concentrations. Pharmacokinetic data were obtained in subjects with multiple sclerosis and healthy volunteers.
Absorption
The median Tmax of MMF is 2-2.5 hours. The peak plasma concentration (Cmax) and overall exposure (AUC) increased approximately dose proportionally in the dose range studied (120 mg to 360 mg). Following administration of TECFIDERA 240 mg twice a day with food, the mean Cmax of MMF was 1.87 mg/L and AUC was 8.21 mg.hr/L in MS patients.
A high-fat, high-calorie meal did not affect the AUC of MMF but decreased its Cmax by 40%. The Tmax was delayed from 2.0 hours to 5.5 hours. In this study, the incidence of flushing was reduced by approximately 25% in the fed state.
Distribution
The apparent volume of distribution of MMF varies between 53 and 73 L in healthy subjects. Human plasma protein binding of MMF is 27-45% and independent of concentration.
Metabolism
In humans, dimethyl fumarate is extensively metabolized by esterases, which are ubiquitous in the gastrointestinal tract, blood, and tissues, before it reaches the systemic circulation. Further metabolism of MMF occurs through the tricarboxylic acid (TCA) cycle, with no involvement of the cytochrome P450 (CYP) system. MMF, fumaric and citric acid, and glucose are the major metabolites in plasma.
Elimination
Exhalation of CO2 is the primary route of elimination, accounting for approximately 60% of the TECFIDERA dose. Renal and fecal elimination are minor routes of elimination, accounting for 16% and 1% of the dose respectively. Trace amounts of unchanged MMF were present in urine.
The terminal half-life of MMF is approximately 1 hour and no circulating MMF is present at 24 hours in the majority of individuals. Accumulation of MMF does not occur with multiple doses of TECFIDERA.
Specific Populations
Body weight, gender, and age do not require dosage adjustment.
No studies have been conducted in subjects with hepatic or renal impairment. However, neither condition would be expected to affect exposure to MMF and therefore no dosage adjustment is necessary.
Drug Interaction Studies
No potential drug interactions with dimethyl fumarate or MMF were identified in in vitro CYP inhibition and induction studies, or in P-glycoprotein studies. Single doses of interferon beta-1a or glatiramer acetate did not alter the pharmacokinetics of MMF. Aspirin, when administered approximately 30 minutes before TECFIDERA, did not alter the pharmacokinetics of MMF.
Oral Contraceptives
The coadministration of dimethyl fumarate with a combined oral contraceptive (norelgestromin and ethinyl estradiol) did not elicit any relevant effects in oral contraceptives exposure. No interaction studies have been performed with oral contraceptives containing other progestogens.
Vaccines
A randomized, open-label study examined the concomitant use of TECFIDERA and several non-live vaccines in adults 27-55 years of age with relapsing forms of MS (38 subjects undergoing treatment with TECFIDERA at the time of vaccination and 33 subjects undergoing treatment with non-pegylated interferon at the time of vaccination). Concomitant exposure to TECFIDERA did not attenuate antibody responses to tetanus toxoid-containing vaccine, pneumococcal polysaccharide, and meningococcal vaccines relative to antibody responses in interferon-treated patients. The impact of these findings on vaccine effectiveness in this patient population is unknown. The safety and effectiveness of live or live-attenuated vaccines administered concomitantly with TECFIDERA have not been assessed.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies of dimethyl fumarate (DMF) were conducted in mice and rats. In mice, oral administration of DMF (25, 75, 200, and 400 mg/kg/day) for up to two years resulted in an increase in nonglandular stomach (forestomach) and kidney tumors: squamous cell carcinomas and papillomas of the forestomach in males and females at 200 and 400 mg/kg/day; leiomyosarcomas of the forestomach at 400 mg/kg/day in males and females; renal tubular adenomas and carcinomas at 200 and 400 mg/kg/day in males; and renal tubule adenomas at 400 mg/kg/day in females. Plasma MMF exposure (AUC) at the highest dose not associated with tumors in mice (75 mg/kg/day) was similar to that in humans at the recommended human dose (RHD) of 480 mg/day.
In rats, oral administration of DMF (25, 50, 100, and 150 mg/kg/day) for up to two years resulted in increases in squamous cell carcinomas and papillomas of the forestomach at all doses tested in males and females, and in testicular interstitial (Leydig) cell adenomas at 100 and 150 mg/kg/day. Plasma MMF AUC at the lowest dose tested was lower than that in humans at the RHD.
Mutagenesis
Dimethyl fumarate (DMF) and monomethyl fumarate (MMF) were not mutagenic in the in vitro bacterial reverse mutation (Ames) assay. DMF and MMF were clastogenic in the in vitro chromosomal aberration assay in human peripheral blood lymphocytes in the absence of metabolic activation. DMF was not clastogenic in the in vivo micronucleus assay in the rat.
Impairment of Fertility
In male rats, oral administration of DMF (75, 250, and 375 mg/kg/day) prior to and throughout the mating period had no effect on fertility; however, increases in non-motile sperm were observed at the mid and high doses. The no-effect dose for adverse effects on sperm is similar to the recommended human dose (RHD) of 480 mg/day on a body surface area (mg/m2) basis.
In female rats, oral administration of DMF (20, 100, and 250 mg/kg/day) prior to and during mating and continuing to gestation day 7 caused disruption of the estrous cycle and increases in embryolethality at the highest dose tested. The highest dose not associated with adverse effects (100 mg/kg/day) is twice the RHD on a mg/m2 basis.
Testicular toxicity (germinal epithelial degeneration, atrophy, hypospermia, and/or hyperplasia) was observed at clinically relevant doses in mice, rats, and dogs in subchronic and chronic oral toxicity studies of DMF, and in a chronic oral toxicity study evaluating a combination of four fumaric acid esters (including DMF) in rats.
13.2 Animal Toxicology and/or Pharmacology
Kidney toxicity was observed after repeated oral administration of dimethyl fumarate (DMF) in mice, rats, dogs, and monkeys. Renal tubule epithelia regeneration, suggestive of tubule epithelial injury, was observed in all species. Renal tubular hyperplasia was observed in rats with dosing for up to two years. Cortical atrophy and interstitial fibrosis were observed in dogs and monkeys at doses above 5 mg/kg/day. In monkeys, the highest dose tested (75 mg/kg/day) was associated with single cell necrosis and multifocal and diffuse interstitial fibrosis, indicating irreversible loss of renal tissue and function. In dogs and monkeys, the 5 mg/kg/day dose was associated with plasma MMF exposures less than or similar to that in humans at the recommended human dose (RHD).
A dose-related increase in incidence and severity of retinal degeneration was observed in mice following oral administration of DMF for up to two years at doses above 75 mg/kg/day, a dose associated with plasma MMF exposure (AUC) similar to that in humans at the RHD.
14. Clinical Studies
The efficacy and safety of TECFIDERA were demonstrated in two studies (Studies 1 and 2) that evaluated TECFIDERA taken either twice or three times a day in patients with relapsing-remitting multiple sclerosis (RRMS). The starting dose for TECFIDERA was 120 mg twice or three times a day for the first 7 days, followed by an increase to 240 mg twice or three times a day. Both studies included patients who had experienced at least 1 relapse over the year preceding the trial or had a brain Magnetic Resonance Imaging (MRI) scan demonstrating at least one gadolinium-enhancing (Gd+) lesion within 6 weeks of randomization. The Expanded Disability Status Scale (EDSS) was also assessed and patients could have scores ranging from 0 to 5. Neurological evaluations were performed at baseline, every 3 months, and at the time of suspected relapse. MRI evaluations were performed at baseline, month 6, and year 1 and 2 in a subset of patients (44% in Study 1 and 48% in Study 2).
Study 1: Placebo-Controlled Trial in RRMS
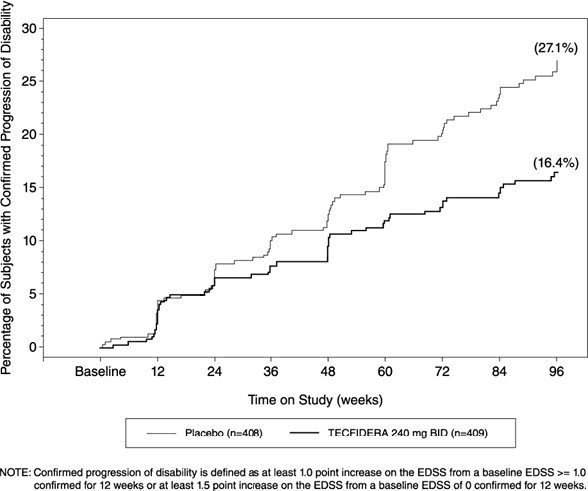
Study 1 was a 2-year randomized, double-blind, placebo-controlled study in 1234 patients with RRMS. The primary endpoint was the proportion of patients relapsed at 2 years. Additional endpoints at 2 years included the number of new or newly enlarging T2 hyperintense lesions, number of new T1 hypointense lesions, number of Gd+ lesions, annualized relapse rate (ARR), and time to confirmed disability progression. Confirmed disability progression was defined as at least a 1 point increase from baseline EDSS (1.5 point increase for patients with baseline EDSS of 0) sustained for 12 weeks.
Patients were randomized to receive TECFIDERA 240 mg twice a day (n=410), TECFIDERA 240 mg three times a day (n=416), or placebo (n=408) for up to 2 years. The median age was 39 years, median time since diagnosis was 4 years, and median EDSS score at baseline was 2. The median time on study drug for all treatment arms was 96 weeks. The percentages of patients who completed 96 weeks on study drug per treatment group were 69% for patients assigned to TECFIDERA 240 mg twice a day, 69% for patients assigned to TECFIDERA 240 mg three times a day and 65% for patients assigned to placebo groups.
TECFIDERA had a statistically significant effect on all of the endpoints described above and the 240 mg three times daily dose showed no additional benefit over the TECFIDERA 240 mg twice daily dose. The results for this study (240 mg twice a day vs. placebo) are shown in Table 2 and Figure 1.
| TECFIDERA
240 mg BID |
Placebo |
P-value |
|
|---|---|---|---|
| Clinical Endpoints | N=410 | N=408 | |
| Proportion relapsing (primary endpoint) Relative risk reduction | 27% 49% | 46% | <0.0001 |
| Annualized relapse rate | 0.172 | 0.364 | <0.0001 |
| Relative reduction | 53% | ||
| Proportion with disability progression | 16% | 27% | 0.0050 |
| Relative risk reduction | 38% | ||
| MRI Endpoints | N=152 | N=165 | |
| Mean number of new or newly enlarging | 2.6 | 17 | <0.0001 |
| T2 lesions over 2 years | |||
| | |||
| Percentage of subjects with no new or newly enlarging lesions | 45% | 27% | |
| Number of Gd+ lesions at 2 years | 0.1 (0) | 1.8 (0) | |
| Mean (median) | |||
| Percentage of subjects with | |||
| 0 lesions | 93% | 62% | |
| 1 lesion | 5% | 10% | |
| 2 lesions | <1% | 8% | |
| 3 to 4 lesions | 0 | 9% | |
| 5 or more lesions | <1% | 11% | |
| Relative odds reduction | 90% | <0.0001 | |
| (percentage) | |||
| Mean number of new T1 hypointense | 1.5 | 5.6 | <0.0001 |
| lesions over 2 years |
Figure 1: Time to 12-Week Confirmed Progression of Disability (Study 1)
Study 2: Placebo-Controlled Trial in RRMS
Study 2 was a 2-year multicenter, randomized, double-blind, placebo-controlled study that also included an open-label comparator arm in patients with RRMS. The primary endpoint was the annualized relapse rate at 2 years. Additional endpoints at 2 years included the number of new or newly enlarging T2 hyperintense lesions, number of T1 hypointense lesions, number of Gd+ lesions, proportion of patients relapsed, and time to confirmed disability progression as defined in Study 1.
Patients were randomized to receive TECFIDERA 240 mg twice a day (n=359), TECFIDERA 240 mg three times a day (n=345), an open-label comparator (n=350), or placebo (n=363) for up to 2 years. The median age was 37 years, median time since diagnosis was 3 years, and median EDSS score at baseline was 2.5. The median time on study drug for all treatment arms was 96 weeks. The percentages of patients who completed 96 weeks on study drug per treatment group were 70% for patients assigned to TECFIDERA 240 mg twice a day, 72% for patients assigned to TECFIDERA 240 mg three times a day and 64% for patients assigned to placebo groups.
TECFIDERA had a statistically significant effect on the relapse and MRI endpoints described above. There was no statistically significant effect on disability progression. The TECFIDERA 240 mg three times daily dose resulted in no additional benefit over the TECFIDERA 240 mg twice daily dose. The results for this study (240 mg twice a day vs. placebo) are shown in Table 3.
| TECFIDERA
240 mg BID |
Placebo |
P-value |
|
|---|---|---|---|
| Clinical Endpoints | N=359 | N=363 | |
| Annualized relapse rate | 0.224 | 0.401 | <0.0001 |
| Relative reduction | 44% | ||
| Proportion relapsing | 29% | 41% | 0.0020 |
| Relative risk reduction | 34% | ||
| Proportion with disability progression | 13% | 17% | 0.25 |
| Relative risk reduction | 21% | ||
| MRI Endpoints | N=147 | N=144 | |
| Mean number of new or newly enlarging | 5.1 | 17.4 | <0.0001 |
| T2 lesions over 2 years | |||
| Percentage of subjects with no new or | 27% | 12% | |
| newly enlarging lesions | |||
| Number of Gd+ lesions at 2 years | |||
| Mean (median) | 0.5 (0.0) | 2.0 (0.0) | |
| Percentage of subjects with | |||
| 0 lesions | 80% | 61% | |
| 1 lesion | 11% | 17% | |
| 2 lesions | 3% | 6% | |
| 3 to 4 lesions | 3% | 2% | |
| 5 or more lesions | 3% | 14% | |
| Relative odds reduction | 74% | <0.0001 |
|
| (percentage) | |||
| Mean number of new T1 hypointense | 3.0 | 7.0 | <0.0001 |
| lesions over 2 years |
16. How is Tecfidera supplied
TECFIDERA is available as hard gelatin delayed-release capsules in two strengths containing either 120 mg or 240 mg of dimethyl fumarate. The green and white 120 mg capsules are printed with “BG-12 120 mg” in black ink. The green 240 mg capsules are printed with “BG-12 240 mg” in black ink. TECFIDERA is available as follows:
30-day Starter Pack, (NDC 64406-007-03):
- 7-day bottle 120 mg capsules, quantity 14
- 23-day bottle 240 mg capsules, quantity 46
120 mg capsules:
- 7-day bottle of 14 capsules (NDC 64406-005-01)
240 mg capsules:
- 30-day bottle of 60 capsules (NDC 64406-006-02)
Store at 15°C to 30°C (59 to 86°F). Protect the capsules from light. Store in original container.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Dosage
Inform patients that they will be provided two strengths of TECFIDERA when starting treatment: 120 mg capsules for the 7 day starter dose and 240 mg capsules for the maintenance dose, both to be taken twice daily. Inform patients to swallow TECFIDERA capsules whole and intact. Inform patients to not crush, chew, or sprinkle capsule contents on food. Inform patients that TECFIDERA can be taken with or without food [see Dosage and Administration (2.1)].
Anaphylaxis and Angioedema
Advise patients to discontinue TECFIDERA and seek medical care if they develop signs and symptoms of anaphylaxis or angioedema [see Warnings and Precautions (5.1)].
Progressive Multifocal Leukoencephalopathy
Inform patients that progressive multifocal leukoencephalopathy (PML) has occurred in patients who received TECFIDERA. Inform the patient that PML is characterized by a progression of deficits and usually leads to death or severe disability over weeks or months. Instruct the patient of the importance of contacting their doctor if they develop any symptoms suggestive of PML. Inform the patient that typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes [see Warnings and Precautions (5.2)].
Herpes Zoster and Other Serious Opportunistic Infections
Inform patients that herpes zoster and other serious opportunistic infections have occurred in patients who received TECFIDERA. Instruct the patient of the importance of contacting their doctor if they develop any signs or symptoms associated with herpes zoster or other serious opportunistic infections [see Warnings and Precautions (5.3)].
Lymphocyte Counts
Inform patients that TECFIDERA may decrease lymphocyte counts. A blood test should be obtained before they start therapy. Blood tests are also recommended after 6 months of treatment, every 6 to 12 months thereafter, and as clinically indicated [see Warnings and Precautions (5.3), Adverse Reactions (6.1)].
Liver Injury
Inform patients that TECFIDERA may cause liver injury. Instruct patients treated with TECFIDERA to report promptly any symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice. A blood test should be obtained before patients start therapy and during treatment, as clinically indicated [see Warnings and Precautions (5.4)].
Flushing
Inform patients that flushing is one of the most common reactions, especially at the initiation of therapy, and may decrease over time. Advise patients to contact their healthcare provider if they experience persistent and/or severe flushing. Advise patients experiencing flushing that taking TECFIDERA with food or taking a non-enteric coated aspirin prior to taking TECFIDERA may help [see Adverse Reactions (6.1)].
Gastrointestinal (GI) Events
Inform patients that GI events (abdominal pain, diarrhea, and nausea) are some of the most common adverse reactions, especially at the initiation of therapy, and may decrease over time. Some patients may experience more severe GI events. Advise patients to immediately contact their healthcare provider and discontinue TECFIDERA if they experience gastrointestinal bleeding (e.g., rectal bleeding, bloody diarrhea, hematemesis) or other serious gastrointestinal adverse events (e.g., severe abdominal pain, severe vomiting and/or diarrhea) [see Warnings and Precautions (5.7)].
PPI-41347-18
Manufactured for:
Biogen Inc.
Cambridge, MA 02142
TECFIDERA is a registered trademark of Biogen.
© Biogen 2013 - 2024
|
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 03/2024 |
|
PPI-41347-3 |
| Patient Information
TECFIDERA® (tek" fi de' rah) (dimethyl fumarate) delayed-release capsules |
What is TECFIDERA?
|
Who should not take TECFIDERA?
|
Before taking and while you take TECFIDERA, tell your doctor if you have or have had:
|
Tell your doctor if you are:
|
How should I take TECFIDERA?
|
| What are the possible side effects of TECFIDERA?
TECFIDERA may cause serious side effects including:
|
The most common side effects of TECFIDERA include:
|
| These are not all the possible side effects of TECFIDERA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. For more information go to dailymed.nlm.nih.gov. |
General Information about the safe and effective use of TECFIDERA
|
| What are the ingredients in TECFIDERA?
Active ingredient: dimethyl fumarate Inactive ingredients: microcrystalline cellulose, silicified microcrystalline cellulose, croscarmellose sodium, talc, silica colloidal silicon dioxide, magnesium stearate, triethyl citrate, methacrylic acid copolymer - Type A, methacrylic acid copolymer dispersion, simethicone (30% emulsion), sodium lauryl sulphate, and polysorbate 80. Capsule Shell: gelatin, titanium dioxide, FD&C blue 1; brilliant blue FCF, yellow iron oxide and black iron oxide. Manufactured for: Biogen Inc., Cambridge, MA 02142, www.TECFIDERA.com or call 1-800-456-2255 |
Principal Display Panel - 240 mg Capsules: Box Label
NDC 64406-006-02
60 capsules
Tecfidera®
(dimethyl fumarate)
delayed-release capsules
240 mg
Dispense in original Package.
Swallow capsule whole.
Rx only
Principal Display Panel - 120 mg Capsules: Box Label
NDC 64406-005-01
14 capsules
Tecfidera®
(dimethyl fumarate)
delayed-release capsules
120 mg
Dispense in original Package.
Swallow capsule whole.
Rx only
Principal Display Panel - 120 mg Capsules: No Charge Box Label
NDC 64406-005-08
14 capsules
Tecfidera®
(dimethyl fumarate)
delayed-release capsules
120 mg
Dispense in original Package.
Swallow capsule whole.
Rx only
Principal Display Panel - 120 mg Capsules: Sample Box Label
NDC 64406-005-10
14 capsules
Tecfidera®
(dimethyl fumarate)
delayed-release capsules
120 mg
Dispense in original Package.
Swallow capsule whole.
Rx only
Principal Display Panel - Starter Pack: Box Label
NDC 64406-007-03
30-Day Starter Pack
Tecfidera®
(dimethyl fumarate)
delayed-release capsules
Package Contents:
120 mg Starter Dose
One bottle containing 14
capsules of 120 mg each
240 mg Regular Dose
One bottle containing 46
capsules of 240 mg each
Dispense in Original Package.
See back panel for dosage and administration
instructions for use.
| TECFIDERA
dimethyl fumarate kit |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| TECFIDERA
dimethyl fumarate capsule |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| TECFIDERA
dimethyl fumarate capsule |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Biogen Inc. (121376230) |
Frequently asked questions
- Is Tecfidera an immunosuppressant?
- How do Vumerity and Tecfidera compare for multiple sclerosis (MS)?
- Can I stop taking Tecfidera? What happens if I do?
- What causes flushing with Tecfidera?
- Can you drink alcohol while taking Tecfidera?
- Gilenya vs Tecfidera. How do they compare?
- How long can you take Tecfidera?
- Can it cause stomach damage?
- Does Tecfidera cause weight gain or loss?
More about Tecfidera (dimethyl fumarate)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (98)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Generic availability
- Support group
- FDA approval history
- Drug class: selective immunosuppressants
- Breastfeeding
- En español